
The FDA has approved the first female libido drug for millions of additional women, reopening a debate many thought was settled years ago. Addyi was once hailed as a historic breakthrough, then quietly fizzled after disappointing sales, safety concerns, and insurer resistance.
Now, a nationwide expansion targets postmenopausal women, raising a critical question: has anything truly changed, or is history about to repeat itself. Let’s look into this deeper.
What Is Actually Happening Now

On December 15, the FDA expanded approval of Addyi to include postmenopausal women up to age 65, dramatically widening its potential audience nationwide. The decision revived a drug many investors and physicians had written off years earlier. Supporters call it long overdue progress in women’s health, while critics see a familiar experiment restarting under new branding. The timing reveals why this moment matters more than it appears.
How Addyi Is Supposed To Work
Addyi, also known as flibanserin, is taken daily at bedtime and works on brain chemistry rather than blood flow. It reduces serotonin while increasing dopamine and norepinephrine to influence sexual motivation, according to DrugBank clinical data. Unlike male erectile dysfunction drugs, it targets desire, not performance. That distinction shaped both its promise and its problems, which became clear once regulators examined the risks.
Years Of Rejection Before Approval
Boehringer Ingelheim first submitted flibanserin to the FDA in 2010, but regulators rejected it over marginal effectiveness and safety concerns. A second rejection followed in 2013 after additional studies, with the FDA demanding more data on driving impairment and alcohol interactions. These setbacks reflected deep skepticism. Yet something shifted by August 18, 2015, when the agency reconsidered under intense external pressure.
When Advocacy Changed The Conversation
The Even the Score campaign framed Addyi as a women’s rights issue, mobilizing major advocacy groups to pressure the FDA, according to a Hastings Center investigation. Organizations testified as grassroots voices while receiving undisclosed funding from Sprout Pharmaceuticals. Many members were unaware of the financial ties. This blending of activism and marketing altered the tone of the debate, reshaping how regulators viewed the approval decision.
A Historic Yes With Heavy Conditions

On August 18, 2015, the FDA approved Addyi—the first prescription drug for female sexual desire disorder—but with strict safeguards. Dr. Hylton V. Joffe, director of the FDA’s Division of Bone, Reproductive and Urologic Products, noted “considerable disagreement” in the 18-6 advisory committee vote, FDA records show. Approval required REMS certification. Just two days later, Sprout sold the drug for $1 billion, betting investors would recover.
Sales Expectations Fell Apart Fast
Wall Street predicted $100 million to $300 million in annual sales, but reality hit quickly. By November 2015, just 227 prescriptions had been written nationwide. Fewer than 1% of primary care physicians completed the required REMS training. Awareness stalled, access narrowed, and enthusiasm faded. By 2016, sales reached roughly $11 million, exposing a mismatch between projections and patient demand that could not be ignored.
The Billion Dollar Bet Unraveled

Valeant Pharmaceuticals bought Addyi for $1 billion just days after FDA approval, expecting a blockbuster. Instead, by 2017, Valeant sold the drug back to Sprout’s founders for a fraction of that price, signaling a massive write-down. The reversal stunned investors and confirmed commercial failure.
However, within Sprout’s offices, executives were already plotting a different strategy: instead of fighting for premenopausal women, why not try older women?
What The Clinical Data Really Shows

Clinical trials painted a sobering picture. In the SNOWDROP study, postmenopausal women taking Addyi reported 0.69 additional sexually satisfying events per month compared to placebo. A 2024 meta-analysis of 8,000 women found an increase of just 0.37 events monthly. Researchers concluded there was no clear evidence of effectiveness. These modest gains undermined claims of a transformational therapy.
Side Effects Proved Hard To Ignore

Addyi’s adverse effects dramatically exceeded placebo rates: 11.4% experienced dizziness versus 2.2% on placebo (5-fold increase), 11.2% reported somnolence versus 2.9% (4-fold increase), and 10.4% felt nausea versus 3.9%, according to Addyi clinical trial data. Overall, 13% of women discontinued use due to adverse reactions compared to 6% on placebo.
Beyond these common effects, a boxed warning documented dangerously low blood pressure and fainting risks when combining the drug with alcohol—a restriction that would define the drug’s fate.
Alcohol Became The Deal Breaker

The boxed warning states that combining Addyi with alcohol risks severe hypotension and syncope (fainting). FDA regulations require women to wait at least 2 hours after consuming 1–2 drinks before taking Addyi, skip the dose if they’ve had 3+ drinks, and cannot drink until the following morning, according to FDA prescribing information.
For a libido drug, this is catastrophic. Intimacy often accompanies wine, cocktails, or social drinking—contexts where women can’t safely use Addyi. The restriction isn’t a minor inconvenience; it’s architectural proof the drug conflicts with real life.
Insurance Barriers Hit Women Harder

Insurance companies systematically denied Addyi claims while approving Viagra, according to CNN reporting and healthcare industry analysis. Blue Shield of California required psychiatric evaluation before covering Addyi—a prerequisite not imposed on men seeking erectile dysfunction drugs.
Commercial insurers demanded women “fail” cheaper unapproved alternatives first (off-label antidepressants) before considering Addyi—a step therapy requirement absent for male ED medications. Dr. Irwin Goldstein, a sexual health expert and consultant to both Addyi and Viagra manufacturers, called this “shocking” and “discriminatory.” The structural bias was undeniable.
A Rival Drug Met The Same Fate
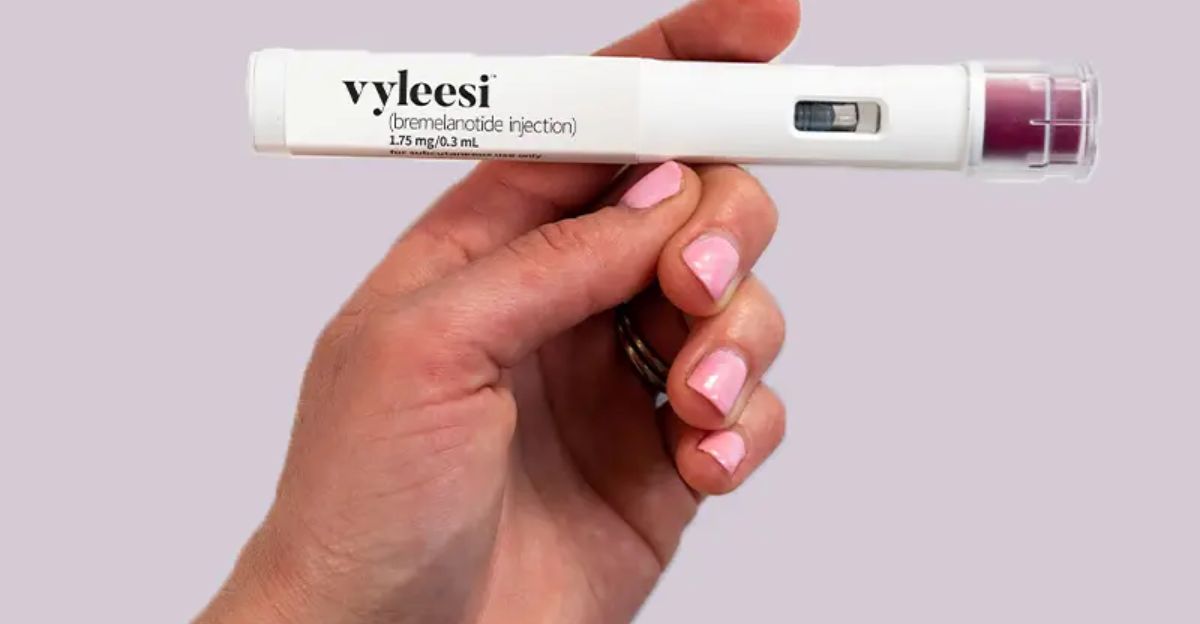
The FDA approved Vyleesi (bremelanotide), an on-demand injection for female HSDD, on June 21, 2019, positioning it as Addyi’s alternative, according to FDA approval records. Vyleesi worked on different neurological targets and didn’t require daily dosing.
Yet Vyleesi also struggled commercially, generating only $12.5 million in annual sales by fiscal year 2023—far short of the $108 million Barclays predicted for 2025, according to Palatin Technologies fiscal reports. By 2024, Palatin sold Vyleesi to Cosette Pharmaceuticals for a $12 million upfront payment, signaling the asset would “never make significant market impact.”
Why Sprout Targeted Older Women

After premenopausal women rejected Addyi, Sprout’s strategy shifted: target postmenopausal women aged 50–65. The biological rationale seemed solid—falling estrogen and testosterone during menopause trigger hormonal upheaval, potentially making HSDD more prevalent and neurologically distinct, according to NCBI menopause literature.
The FDA granted Priority Review to the postmenopausal indication in July, signaling recognition of an “unmet medical need.” But insiders understood the real motive: premenopausal sales had collapsed, and executives were desperate to find a demographic that might embrace a decade-old drug rejected by their mothers’ generation.
How The Key Trial Was Designed
The SNOWDROP trial enrolled 949 postmenopausal women (468 on Addyi, 481 on placebo) over 24 weeks, creating a homogeneous cohort: all postmenopausal for at least 1 year, all in stable heterosexual relationships, according to Addyi HCP trial design. This standardization was methodologically sound—it controlled variables.
But it also ensured participants weren’t representative of real-world postmenopausal women, many of whom struggle with relationship stress, single status, or medical comorbidities that complicate HSDD diagnosis. The trial delivered what Sprout needed: a statistically significant (though clinically modest) result justifying expansion.
International Approval Came First
Health Canada approved Addyi for postmenopausal women in 2021—4 years before the U.S. expansion—according to FDA and Health Canada approval records. This lag raises questions: Did the FDA move slower due to lingering concerns from 2015’s controversial approval? Or did international regulatory confidence eventually sway U.S. decision-makers?
The Canadian precedent mattered because it showed another developed nation’s regulator had already deemed the expansion appropriate. Yet the U.S. FDA waited until December 15, 2025, to grant the same approval, suggesting institutional caution or political sensitivity around the drug’s legacy.
The Price Shock Few Discuss

Addyi costs approximately $1,170–$1,185 per month at retail price ($14,040 annually), according to GoodRx and SingleCare pricing data. Even with manufacturer copay assistance programs bundling 3 months at the price of 2, patients rarely see coverage approaching Viagra’s $10–15 per pill ($50–150 monthly for generics).
Medicare Part D coverage varies by plan with potential prior authorization requirements. Medicaid differs state-by-state. For uninsured women, the drug is prohibitively expensive—creating an ironic outcome where expansion theoretically reaches 15–25 million women, yet only affluent women with excellent insurance actually access it.
The Official Expansion Announcement

The FDA approved Addyi for postmenopausal women up to age 65 on December 15, 2025, expanding access from premenopausal women to an estimated 15–25 million additional American women, according to FDA approval statements and press releases.
Sprout Pharmaceuticals’ CEO Cindy Eckert stated: “This reflects a decade of persistent work with the FDA to fundamentally change how women’s sexual health is understood and prioritized,” according to company statements on December 15, 2025.
The rhetoric echoed 2015’s feminist framing. Yet this time, the company had learned from past mistakes—the messaging was more muted, acknowledging the drug’s limitations rather than overselling.
Are The Same Problems Still There?

Sprout faces the same obstacles that crushed Addyi’s first launch: REMS certification requirements limiting prescriber adoption (fewer physicians certified in 2025 than in 2015), insurance barriers more entrenched than ever, and a $1,170 monthly price tag that guarantees access remains limited to the wealthy.
The alcohol warning persists. Side effects remain unchanged. Efficacy is still modest. Clinical trials showed that even in the controlled postmenopausal cohort, improvement was marginal. What has changed is only the patient demographic. Everything else—the barriers, the skepticism, the modest benefits—remains identical to the disaster of 2015.
The Bigger Gender Health Gap

The FDA has approved more than 20 drugs for male sexual dysfunction but only 2 for female dysfunction. Research funding and medical training heavily favor male conditions. The imbalance reflects market incentives rather than scientific impossibility. Addyi’s expansion does little to change that reality unless insurers, physicians, and patients shift behavior. Without systemic change, progress may remain symbolic.
A Second Chance With Unclear Demand

The expansion theoretically unlocks access for millions of postmenopausal women seeking treatment for HSDD. Yet the obstacles remain formidable: insurance denials, REMS prescriber barriers, $1,170 monthly costs, alcohol restrictions, and modest efficacy that delivers less than 1 additional satisfying sexual event per month. Vyleesi’s failure suggests female libido drugs struggle not because of scientific doubt but because real-world barriers outweigh benefits.
Sprout’s 50-person company faces an uphill battle marketing a decade-old drug with a complicated history to a demographic already skeptical of pharmaceutical solutions. The expansion is real. Success, however, remains uncertain.
Sources:
“Historic First in Women’s Sexual Health: FDA Grants Approval for Addyi (flibanserin) for Postmenopausal Women.” PR Newswire, December 15, 2025.
“Flibanserin.” Wikipedia (citing peer-reviewed clinical data), accessed December 2025.
“Addyi Alcohol Warnings Have Not Been Revised, Says FDA.” EMPR, September 11, 2019.
“FDA OKs Libido-Boosting Pill for Women Who Have Gone Through Menopause.” ABC News, December 15, 2025.
“‘Female Viagra’ Keeps Alcohol Black Box Warning Amid Safety Concerns.” BiopharmaDive, April 11, 2019.
“Behind the Scenes at the FDA: Why They Ultimately Approved Addyi.” BiopharmaDive (citing FDA institutional records and advisory committee minutes), accessed 2025.
“The Score is Even: Lessons from the Addyi Campaign.” Hastings Center Report, June 27, 2022.